Research Progress
Researchers Developed a Novel NKP Cell Therapy for Cancer treatment
Chinese researchers have developed a de novo pluripotent stem cell (PSC)-derived chimeric antigen receptor NK progenitor (CAR-NKP) cell therapy, offering a promising new strategy for cancer immunotherapy.
CAR-NK cell therapy is considered safe and universally applicable for the treatment of cancer and autoimmune diseases. However, CAR-NK cells demonstrate limited expansion and persistence in vivo, resulting in suboptimal efficacy. As a result, high cell doses and multiple infusions are typically required to achieve durable clinical responses.
A research team led by Prof. WANG Jinyong from the Institute of Zoology of the Chinese Academy of Sciences has developed a CAR-PSC-derived NKP (CAR-iNKP) cell therapy, enabling robust in vivo generation of long-lived CAR-iNK cells. The study, entitled "Pluripotent stem cell-derived CAR-NK progenitor therapy targets minimal residual disease and prevents relapse in leukemia models" was published online in Cell Stem Cell on February 24, 2026.
Prof. WANG's research team has developed an air-liquid interface organoid induction system for the large-scale production of induced NK (iNK) cells from human PSCs (hPSCs). Based on this previous study, the team identified a cell population phenotypically and functionally resembling natural NK progenitor (NKP) cells, termed iNKP cells. The hPSCs were engineered to express CXCR4, generating CXCR4-expressing iNKP (R4-iNKP) cells with enhanced bone marrow niche homing capability. A single low dose of R4-iNKP cells (2 × 105 cells per mouse), administered via tail vein injection into IL-15 humanized B-NDG mice, successfully migrated to the bone marrow niche and differentiated into functional R4-iNK cells within 7–10 days. Encouragingly, the R4-iNK cells persisted in peripheral blood for over 80 days. Furthermore, CAR-R4iNKP cell infusion also resulted in similar long-term in vivo persistence of CAR-R4iNK cells.
The therapeutic potential of CD19- and CD7-targeted CAR-R4iNKP cells was evaluated in human B-ALL and T-ALL animal models. Mice pre-infused with CAR-R4iNKP cells were fully protected against subsequent challenges of CD19+ NALM6 and CD7+ CCRF-CEM tumor cells, respectively.
When combined with chemotherapy, a single low-dose of CAR-R4iNKP cell infusion achieved complete long-term remission in B-ALL and reduced T-ALL relapse by 50%. In contrast, all control groups exhibited 100% tumor relapse. Additionally, CAR-R4iNKP infusion alone also significantly suppressed JeKo-1 lymphoma progression.
Cytokine-induced memory-like NK (MLNK) cells show superior in vivo persistence than conventional NK cells. Compared with PBMC-derived CAR-NK and CAR-MLNK cells, iPSC-derived CAR-R4iNK cells from the same donor exhibited longer persistence in vivo. Notably, only the CAR-R4iNKP group achieved long-term remission, while all mice receiving CAR-NK or CAR-MLNK relapsed.
Thus, CAR-iNKP cells overcome the limited persistence of conventional CAR-NK cells and display superior efficacy in eradicating tumor MRD. This study establishes a novel strategy for tumor immunotherapy with high efficacy, long persistence, and potential cost-effectiveness.
Funding for the research was provided by the National Natural Science Foundation of China and the Ministry of Science and Technology of the People's Republic of China.
DOI: https://doi.org/10.1016/j.stem.2026.01.013
Contact:
WANG Jinyong
Institute of Zoology, Chinese Academy of Sciences
Tel: 86-18002250668
E-mail: wangjinyong@ioz.ac.cn
Web:http://english.ioz.cas.cn/
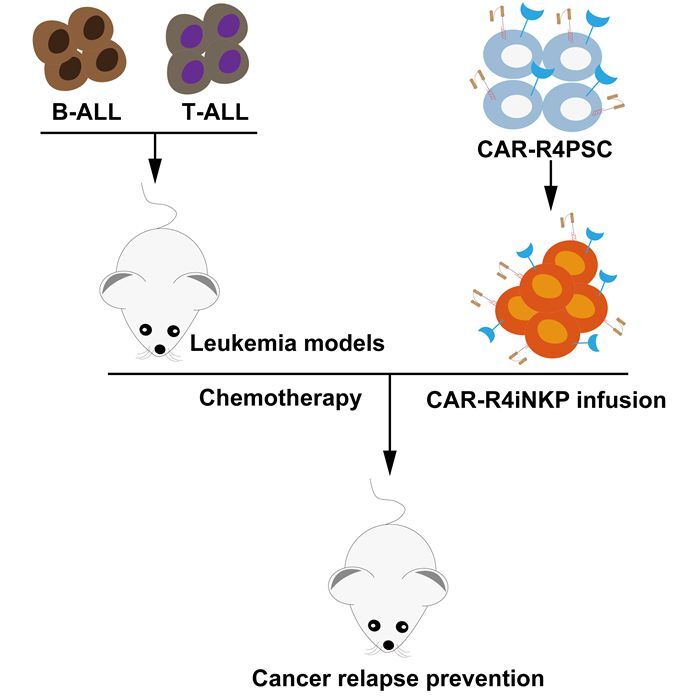
Schematic of CAR-iNKP Cell Therapy (image by WANG Jinyong Lab)



