Research Progress
SPYTAC Enables Safe and Efficient Alzheimer' s Treatment via Lysosomal Targeting
Alzheimer' s disease (AD) affects nearly 57 million people worldwide and is characterized by the accumulation of amyloid-β (Aβ) plaques in the brain. Antibody therapies that target Aβ can slow disease progression, but they often cause brain inflammation and amyloid-related imaging abnormalities (ARIA), including microhemorrhage. Safely removing Aβ without triggering immune side effects has remained a major challenge.
In a study published in Cell on March 4, 2026, a research team led by LI Wei, HU Baoyang, and ZHOU Qi at the Institute of Zoology, Chinese Academy of Sciences, reports a new therapeutic strategy called SPYTAC (synthetic peptide-programmed lysosome-targeting chimeras).
SPYTAC is a fully synthetic peptide platform engineered to cross the blood–brain barrier (BBB) and promote Aβ degradation in both the brain and peripheral tissues. Its key feature is a modular design that links an Aβ-binding peptide to a peptide targeting Low-density lipoprotein receptor-related protein 1 (LRP1). LRP1 is highly expressed on BBB endothelial cells and neurons and naturally mediates molecular transport and lysosomal degradation. By bridging Aβ to LRP1, SPYTAC promotes cellular uptake of Aβ and directs it to lysosomes for breakdown. At the same time, it facilitates transport across the BBB, enabling coordinated clearance from both the central nervous system and the periphery.
In the 5xFAD mouse model of AD, SPYTAC significantly reduced Aβ levels in plasma and brain tissue. It also directly targeted established amyloid plaques and improved learning and memory performance. Because SPYTAC does not contain an Fc fragment, it avoids Fc receptor-mediated immune activation. The study observed reduced neuroinflammation and lower risk of microhemorrhage compared with antibody-based therapies. Beyond AD, SPYTAC represents a programmable platform with broader applications. By replacing the disease-targeting peptide module, the system can potentially be adapted to degrade other pathogenic proteins, such as Tau or α-synuclein. Its fully synthetic nature also offers advantages for scalable manufacturing and potential cost-effective.
This work establishes a new paradigm for targeted protein degradation in the central nervous system. By integrating efficient BBB transport, lysosomal degradation, and an improved safety profile, SPYTAC provides a promising strategy for treating Alzheimer's disease and potentially other protein-driven neurological disorders.
DOI: https://doi.org/10.1016/j.cell.2026.01.034
Contact:
LI Wei
Institute of Zoology, Chinese Academy of Sciences
Tel: 86-10-64807299
Email: liwei@ioz.ac.cn
Web: http://english.ioz.cas.cn/
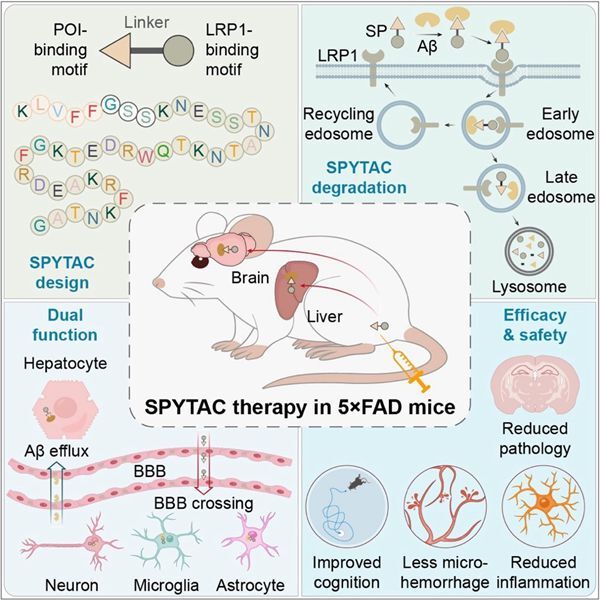
Figure: SPYTAC offers promise for safer and more effective Alzheimer' s Disease treatment
(Image by LI Wei' s Lab)



