Developmental synchrony, resulting from reduced fluctuationinindividual development rates, is the basis of uniform swarming, migration, and sexual maturation. Developmental synchrony is animportant strategy for social animals to keep social relationships and adapt to variable environments.Previous studies ondevelopmental synchrony mainly focused onecology andphysiology, however, the molecular mechanisms underlying developmental synchrony are largely unexplored.
A group led by Professor Kang Le from Institute of Zoology, Chinese Academy of Sciences, studied the molecular mechanisms of developmental synchrony with the model of the migratory locust. The migratory locust exhibitspolyphenism between gregarious and solitarious individuals, with the former displaying more synchronous sexual maturation and migration than the latter. The study found that the egg-hatching time of gregarious locusts was more uniform compared with solitarious locusts and that microRNA-276 (miR-276) was expressed significantly higher in both ovaries and eggs of gregarious locusts than in solitarious locusts. Interestingly, inhibiting miR-276 in gregarious females and overexpressing it in solitarious females, respectively, caused more heterochronic and synchronous hatching of progeny eggs. Moreover, miR-276 directly targeted a transcription coactivator gene, brahma (brm), resulting in its up-regulation. Knockdown of brm not only resulted in asynchronous egg hatching in gregarious locusts but also impaired the miR-276–induced synchronous egg hatching in solitarious locusts. Mechanistically, miR-276 mediated brm activation in a manner that depended on the secondary structure of brm, namely, a stem-loop around the binding site of miR-276. Collectively, the study unravels a mechanism by which miR-276 enhances brm expression to promote developmental synchrony and provide insight into regulation of developmental homeostasis and population sustaining which are closely related to biological synchrony.
Prof. Kang's research has been published online in Proceedings of the National Academy of Sciences of the United States of America (http://www.pnas.org/ on January 4, 2016). The research was supported by Strategic Priority Research Program of the Chinese Academy of Sciences Grant, and National Science Foundation of China Grants.
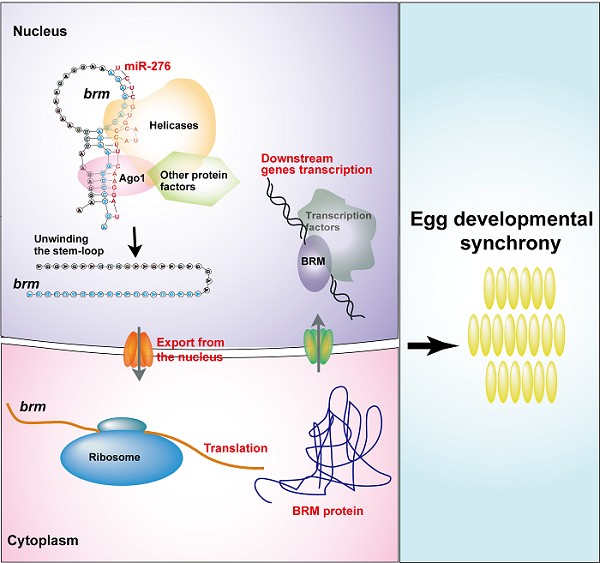
Synchronous development of eggs serves an important basis for the synchrony of hopper development, swarming, migration, and sexual maturation. He et al. found that maternal miR-276 and its target gene brm (a transcription coactivator) promotes developmental synchrony of the embryos. Mechanistically, by recognizing the secondary structure of brm, miR-276 may recruit many protein factors such as helicases to the Ago complex to unwind the stem-loop structure of brm RNA. As a result, miR-276 promotes the nuclear exportation and translation processes of brm RNA. As an important transcriptional coactivator for the homeotic genes expression in early embryonic development, BRM is essential for the stable expression of downstream genes. Thus, miR-276-mediated upregulation of brm promotes the developmental synchrony of the eggs in the locusts. The study provides a previously unidentified mechanism by which miRNA promotes the expression of its target and provide an important cue for the regulation of egg-hatching synchrony by noncoding RNAs.

