The eukaryotic cell has an elaborate internal membrane system, including a number of functionally distinct membrane-bounded compartments (organelles). The transport and localization of organelles within a eukaryotic cell are dependent on a complex network of protein filaments called the cytoskeleton and the associated proteins, including actin filaments and myosins. Myosin is a molecular motor which has ATPase activity and is able to convert the energy from ATP into mechanical movement along actin filaments. To transport cargo efficiently and prevent the futile hydrolysis of ATP, the motor activity of myosin must be tightly regulated.
Unconventional myosin V is one of the most ancient members of the myosin superfamily, distributing from lower eukaryotes, such as yeast, to vertebrates. Vertebrate Myo5a is the best characterized unconventional myosin V in terms of cellular function and molecular regulation. It is now widely accepted the tail-inhibition model for the regulation of Myo5a motor function: the tail of Myo5a is the inhibitory domain which directly interacts the motor domain and inhibits its motor function; high levels of Ca2+ or cargo-binding may reduce the head-tail interaction, thus activating the motor function. However, little was known about the molecular mechanism by which Ca2+ and cargo-binding protein activates the motor function of Myo5a.
In the past few years, the cell motility and muscle contraction research group led by Prof. Xiang-dong Li have made significant progress in understanding the regulation mechanism of Myo5a (JBC 2012; 2016a; 2016b; Sci Rep. 2015; Biochem J. 2015). Importantly, those studies demonstrated that the calmodulin (CaM) bound to the first IQ motif (IQ1) is responsible for Ca2+ regulation of Myo5a. Recently, Xiang-dong Li’s group solved the crystal structure of a truncated Myo5a containing the motor domain and IQ1 in complex with Ca2+-bound calmodulin (Ca2+-CaM), revealing the Ca2+ induced large conformational changes of CaM bound to IQ1 (Figure 1). Biochemical analysis and single molecule assay indicate that CaM continuously associates with the IQ1 during Ca2+ transition (Figure 2). Kinetic analyses show that the binding of CaM to IQ1 increases Ca2+ affinity and substantially changes the kinetics of the Ca2+ transition. Those findings indicate that the IQ1/CaM complex functions as an intact Ca2+ sensor responding to distinct Ca2+ signals. Based on these results, a model for the Ca2+-induced conformational changes of CaM in complex with the Myo5a IQ1 was proposed (Figure 3).
These results were published in Proceedings of the National Academy of Sciences USA (DOI: 10.1073/pnas.1607702113). Dr. Mei Shen, Dr. Ning Zhang and Dr. Sanduo Zheng are the first coauthors and Prof. Xiang-dong Li is the corresponding author of this paper. This work is supported by National Basic Research Program of China Grants and the National Natural Science Foundation of China Grants.
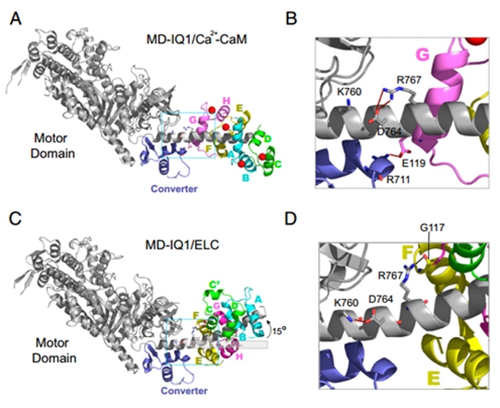
Figure 1. Structural comparison of Myo5a motor domain and IQ1 in complex with Ca2+-CaM and ELC, an equivalent of apo-CaM.
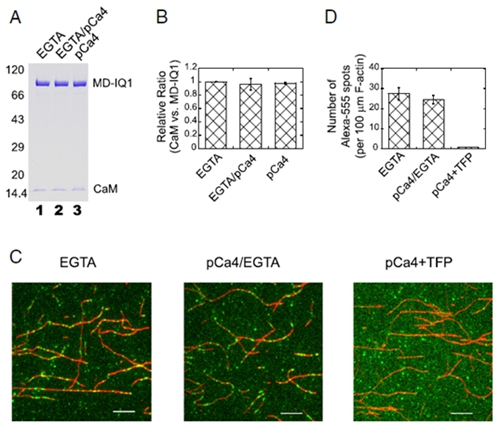
Figure 2. CaM continuously associates with the Myo5a MD-IQ1 during the Ca2+ transition.
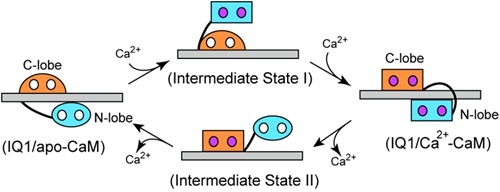
Figure 3. Model for the Ca2+-induced conformational changes of CaM in complex with the Myo5a IQ1.



