Influenza A viruses can be highly contagious pathogens for both humans and several animal species. In support of their efficient replication in the host cells, the viruses interact with a variety of host proteins and hijack cellular responses to antagonize some cellular pathways at different stages. The post-translational conjugation of the Small Ubiquitin-like MOdifier (SUMO) to target proteins (SUMOylation) is an important mechanism to regulate protein activity, stability, cellular localization, and protein-protein interactions. Previous reports indicated that influenza A virus interacted extensively with the cellular SUMOylation system during infection, and NS1, NS2, PB1, NP and M1 proteins were targets of the cellular SUMOylation system. However, whether the host cellular SUMOylation status was affected by influenza A virus infection remains mostly unexplored.
Researchers from the Institute of Zoology of the Chinese Academy of Sciences investigated that the MAPK/ERK pathway could be modified by SUMO1 because the SUMOylation of MEK1 was quickly eliminated after influenza A virus infection.
The scholars found that the host cell MEK1 is as a target of SUMO1 through LC/MS/MS, and further revealed that the enhanced MEK1 SUMOylation can inhibit the infection of the virus, while inhibition of host cell MEK1 SUMOylation facilitated virus propagation.
The scholars also found that the MAPK/ERK pathway is downregulated by MEK1 SUMOylation, which is inhibited by influenza virus infection. Furthermore, membrane accumulation of hemagglutinin promoted MEK1 phosphorylation and gradually abrogated the MEK1 SUMOylation.
Taken together, they report a possible mechanism in which HA may trigger the ERK pathway in influenza A virus-infected cells as the switch from MEK1 SUMOylation to phosphorylation, facilitating virus infection.
This study was supported by National major research and development projects (2016YFD0500300), China Agriculture Research System Poultry-related Science and Technology Innovation Team of Peking (CARS-PSTP).
The study entitled “HA Triggers the switch from MEK1 SUMOylation to Phosphorylation of the ERK pathway in influenza A virus-infected Cells and facilitates its infection” has been published in Frontiers in Cellular and Infection Microbiology on Feb 7th in 2017.(http://journal.frontiersin.org/article/10.3389/fcimb.2017.00027/full ).
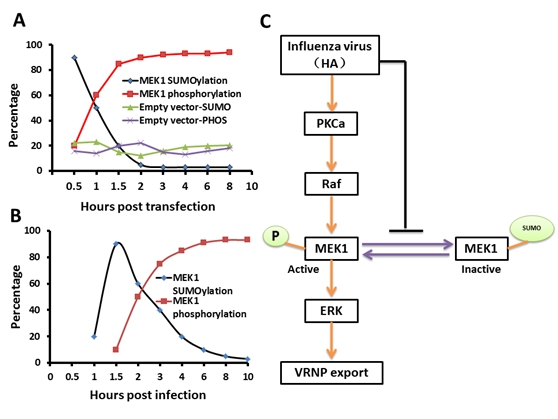
A schematic model of negative regulation of the ERK pathway by MEK1 SUMOylation. The dynamic changes of MEK1-SUMOylation and –phosphorylation show a similar trend for membrane expressing HA(A) and virus infection (B), but the crossover point between treatments for virus infection appears one hour later than when the membrane expresses HA. They propose a model that influenza virus activates the ERK pathway by negatively controlling MEK1 SUMOylation(C). HA of influenza virus is essential for regulation of vRNP export in the ERK pathway through down regulation of MEK1 SUMOylation, namely, HA triggers the switch from MEK1 SUMOylation to phosphorylation of the ERK pathway and facilitates its infection (Image by WANG et. al)
Contact:
HE Hongxuan
Institute of Zoology, Chinese Academy of Sciences (http://english.ioz.cas.cn/)
Chaoyang District, Beijing 100101, P.R.China
Email: hehx@ioz.ac.cn

