Genomic integrity and stability is constantly threatened by DNA damage, which may arise from numerous environmental and intrinsic sources. Failure to repair DNA damage can cause mutations, genomic instability, premature aging, mental retardation, developmental disorders as well as cancers. Cells have evolved the DNA damage response (DDR) pathway to cope with these threats to genome. The DDR is a complex signal transduction pathway which is able to sense damaged DNA and to signal to cell regulating a variety of cellular responses, such as DNA repair, cell cycle arrest, or cell death in case of excessive DNA damage. Chk1, an important effector kinase in the genome surveillance pathways, is activated by ATR-mediated phosphorylation in response to DNA damage or replication stress, thus regulating the above various checkpoint responses.
In collaboration with Dr. Caixia Guo's group in the CAS Key Laboratory of Genomics and Precision Medicine at Beijing Institute of Genomics, Dr. Tie-Shan Tang’s group in the Institute of Zoology at Chinese Academy of Sciences has identified Ataxin-3 (ATX3), mutated in a neurodegenerative disorder Machado-Joseph disease (MJD1, also known as spinocerebellar ataxia type 3, SCA3), as a novel deubiquitinase of Chk1. ATX3 deficiency results in pronounced reduction of Chk1 abundance, compromised DNA damage response, G2/M checkpoint defect and increased sensitivity to replication stress. Mechanistically, ATX3 deubiquitinates both DDB1/CUL4A- and FBXO6-mediated Chk1 polyubiquitination and restrains its degradation in vitro and in vivo, and the deubiquitinase activity of ATX3 is indispensable for its role in stabilizing Chk1. Further experiments revealed a dynamic interaction between ATX3 and Chk1 before and after prolonged replication stress and Ser345 phosphorylation of Chk1, which targets Chk1 for polyubiquitination and degradation, plays an essential role in regulating the interaction between Chk1 and ATX3. Under unperturbed conditions and upon DNA damage, ATX3 interacts with Chk1, and the deubiquitination of Chk1 by ATX3 limits its polyubiquitination and subsequent degradation mediated by Cul1- and Cul4A- containing E3 ligase complexes, thus promoting Chk1 stabilization and further checkpoint signaling and DNA repair. Under prolonged replicative stress, ATX3 dissociates from Chk1, parallel nicely to a stronger association between Chk1 and its E3 ligase, followed by polyubiquitination and proteasomal destruction of Chk1, contributing to checkpoint termination, cell cycle resumption and cell survival.
These new findings demonstrate a critical role of ATX3 in genome integrity/stability maintenance via Chk1 stabilization, which provides new insight into the pathogenic mechanisms of SCA3. SCA3 also manifests dopaminergic neuron degeneration, a main pathologic feature that is similar to Parkinson’s disease, so this work may also shed light on the potential role of DDR pathway defect in dopaminergic neuron degeneration in Parkinson’s.
The study entitled “ATX3 promotes genome integrity by stabilizing Chk1” has been published in Nucleic Acids Research ahead of print on Feb 9, 2017.
Paper link:https://academic.oup.com/nar/article-lookup/doi/10.1093/nar/gkx095
This study was supported by MOST and NFSC.
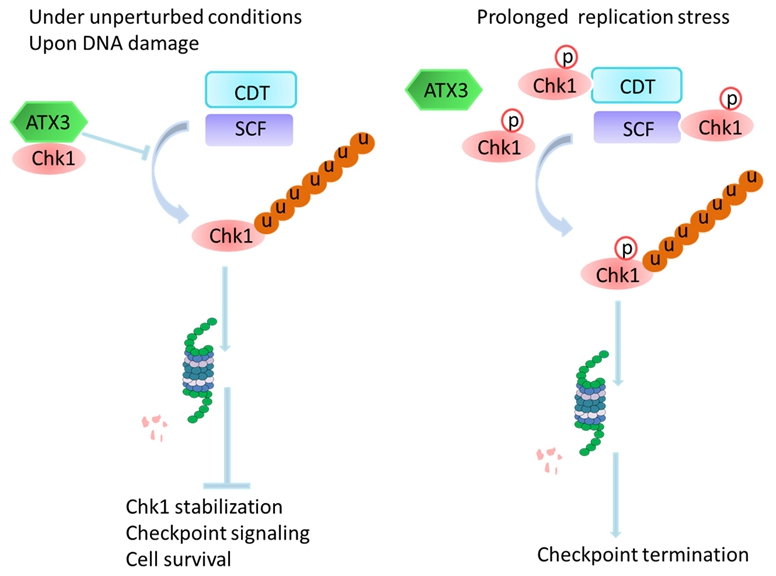
The working model of how ataxin-3 promotes genome integrity by stabilizing Chk1 (Image by TU et. al)
Contact:
TANG Tieshan
Institute of Zoology, Chinese Academy of Sciences (http://english.ioz.cas.cn/)
Chaoyang District, Beijing 100101, P.R.China
Email: tangtsh@ioz.ac.cn

