Congenital hypothyroidism (CH) is caused by thyroid hormone (TH) deficiencies. CH is one of the most prevalent endocrine diseases, occurring in 1 of 1400 to 2800 newborns. Based on anemia often being the first sign of hypothyroidism, accounting for 20% to 60% in hypothyroid patients, the current clinical guidelines recommend evaluating thyroid function in the workup of anemia. Although the most common treatment of hypothyroidism of T4 replacement or T4-T3 combination therapy would bring symptomatic improvement in patients to some extent, some CH patients with anemia remain fail to regain a normal sense of well-being. These observations are suggesting that hematopoietic tissue might be irreversibly impaired from the severe hypothyroidism and the TH supplement alone is not sufficient to fully recover these irreversible lesions. Therefore, the understanding of how TH regulate hematopoiesis might provide a clue of developing optimized treatment protocol for CH.
Animal models are crucial for deciphering disease pathogenesis and developing novel therapeutic agents and treatments. The ideal animal model should resemble not only a human disease phenotype but also its underlying causality. In this circumstance, pigs have anatomical, physiological, and genomic characteristics that are similar to humans, which make them highly suitable for modeling human diseases. More importantly, porcine thyroid extract has been widely used for treatment of CH, and pigs and humans are known to share a high degree of homology in TH metabolism, which implicates that pigs might be suitable animals for the CH studies.
For developing better pig models, the Chinese Swine Mutagenesis Consortium Project which is leading by Dr. Anming Meng and Dr. Qi Zhou, was established and a large-scale screed of mutants in Bama miniature pigs were carried out (eLife, 2017). Among these screen, a mutant line exhibited strikingly weak vitality and nude skin in an autosomal-recessive inheritance pattern was identified. In current study, Dr. Zhao and his team identified this mutant displayed phenotypes of severe CH similar to CH patients. A family-based genome-wide linkage study (GWLS) and whole-genome sequencing (WGS) were performed to map the causative gene and identified a novel recessive c.1226 A>G transition of the dual oxidase 2 (DUOX2) gene responsible for CH. This point mutation resulted in a conserved aspartic acid transition to glycine (D409G, hereafter referred to as DUOX2D409G/D409G). The homozygous DUOX2 knockout pigs created by the CRISPR/Cas9 system exhibited the same phenotype as DUOX2D409G/D409G pigs, further confirms the D409G mutation in DUOX2 is the causative mutation.
Similar to the clinical features of human CH, severe anemia and T lymphopenia were further observed in DUOX2D409G/D409G piglets. By RNA sequencing analysis in the thymuses, KLF9 was identified to be the most dramatically downregulated gene in DUOX2D409G/D409G pigs. Thus we hypothesized that KLF9 may play a vital role in hematopoiesis under the regulation of TH. KLF9 was further verified to be directly regulated in hematopoietic cells by TH in a TH receptor (TR)-dependent manner. Further by using zebrafish model, collaborated with Dr. Feng Liu, we testified that knockdown of klf9 in zebrafish embryos impaired hematopoietic development including erythroid maturation and T lymphopoiesis.
To our knowledge, this study created a CH pig model for the first time and provides the novel mechanism that TH impacts hematopoiesis via KLF9. The current study identifies a novel mechanism for TH-dependent disorders leading to anemia and immunodeficiency, and might provides a clue for developing better treatment plan for human hypothyroidism. In addition, it also provides a new idea for creating new strains with high immunity in pig breeding industry.
The relevant paper entitled "Thyroid hormone regulates hematopoiesis via the TR-KLF9 axis" is published online in Blood on September 25, 2017. This work was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences, the National Transgenic Project of China, the National Natural Science Foundation of China, the National Basic Research Program of China, and the National High Technology Research and Development Program of China.
Manuscript: http://www.bloodjournal.org/content/early/2017/09/22/blood-2017-05-783043
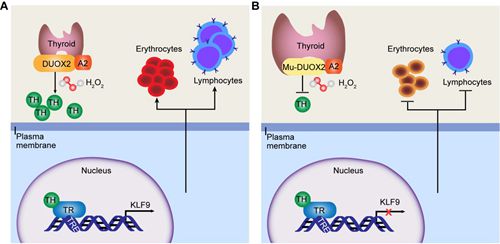
Model of TH regulating hematopoiesis via the TR-KLF9 axis
Contact:
Jianguo Zhao, PhD
State Key Laboratory of Stem Cell and Reproductive Biology, Institute of Zoology, Chinese Academy of Sciences, 1 Beichen West Rd, Chaoyang District, Beijing 100101, China;
Email: zhaojg@ioz.ac.cn

