Hematopoietic stem cells (HSCs) can give rise to all blood lineages, including erythrocyte, myelocyte and lymphocyte, and maintain lifelong hematopoiesis in vertebrates. However, the limited source of matched donor-derived HSCs has greatly hindered their application in treatment of malignant blood diseases, therefore how to induce and expand HSCs in vitro has become one of the most desired approaches in clinic.However, to fulfill this end, a complete understanding of molecular mechanisms of HSC development in vivo is a prerequisite in this field.
In vertebrates, HSCs are generated from a group of specialized endothelial cells (ECs) in the aorta-gonad-mesonephros (AGM) region, named hemogenic endothelium (HE), through a process known as endothelial-to-hematopoietic transition (EHT). Then, HSCs enter into blood circulation and rapidly expand in the fetal liver (human and mouse) or caudal hematopoietic tissue (zebrafish). Finally, HSCs home to the bone marrow (human and mouse) or the kidney (zebrafish) to sustain lifelong hematopoiesis. During these successive processes, HSC development is regulated by a complex of intrinsic and extrinsic regulators spatially and temporally, but the detailed molecular mechanism are still largely unclear, especially for mRNA modifications.
Recently, the Hematopoietic and Cardiovascular Development Group led by Prof. Feng Liu, in collaboration with Prof. Yungui Yang from Beijing Institute of Genomics (BIG) uncovered that the m6A modification regulates the cell fate determination of HSCs in zebrafish (Nature, 2017). Based on this work, Feng Liu’s group continues to explore the regulatory mechanism of m6A modification in mouse HSC development using the conditional knock-out system, in collaboration with Prof. Qi Zhou, Prof. Wei Li from Institute of Zoology and Prof. Yungui Yang from BIG.
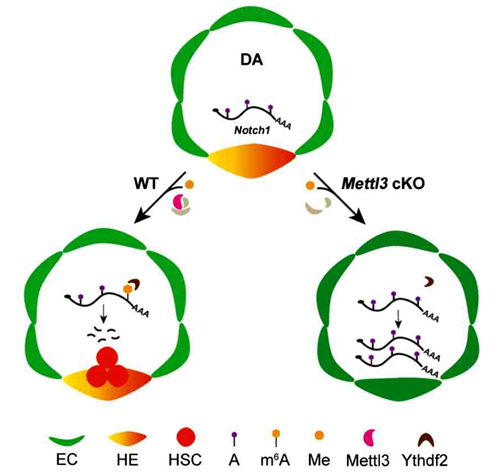
In this study, Feng Liu's group utilized Vec-Cre mice crossed with Mettl3fl/fl mice to deplete the expression of Mettl3 specifically in endothelial cells of mouse AGM region. Mettl3 is a component of the m6A methyltransferase complex, which functions as a RNA modification “writer”. As a result, they found that the level of m6A was significantly reduced in Mettl3-deficient AGM. The phenotype analysis revealed that the development of HSCs was impaired in E10.5 Vec-Cre; Mettl3fl/fl embryo but appeared normally in Vav-Cre; Mettl3fl/flembryo, in which the expression of Mettl3 was specifically deleted in hematopoietic cells. Mechanistically, the RNA-seq data showed a marked upregulation of genes in Notch signaling pathway, especially the expression of Notch1. Meanwhile, m6A-RIP-qPCR assay showed that the enrichment of m6A on Notch1 mRNA was notably decreased in the Vec-Cre; Mettl3fl/fl AGM. These results suggest that the loss of m6A modification leads to the over-activation of Notch signaling pathway, which enhances the arterial identity of ECs in the AGM and blocks the EHT. Additionally, bioinformatics analysis based on the public data showed that the m6A peak near the stop codon of Notch1 mRNA can be recognized by Ythdf2, an m6A reader, suggesting that Ythdf2-mediated Notch1 mRNA decay may be responsible for the observed HSC phenotype. Taken together, endothelial-specific Mettl3 facilitates m6A methylation on Notch1 mRNA to inhibit Notch activity in HE cells, thereby promoting HSC generation through EHT. These results are consistent with our recent findings in zebrafish.
This study demonstrates that epigenetic modification plays an indispensable role in HSC development across vertebrates, as well as provides new insights for HSC regeneration from bench to bedside. This work entitled ‘Endothelial-specific m6A modulates mouse hematopoietic stem and progenitor cell development via Notch signaling’ was published online in Cell Research on Nov. 17, 2017. This work was supported by grants from the National Natural Science Foundation of China, the Ministry of Science and Technology of China and the Strategic Priority Research Program of the Chinese Academy of Sciences, China.
Website: http://www.nature.com/articles/cr2017143
Endothelial-specific m6A modulates mouse hematopoietic stem and progenitor cell development

