Peri-implantation embryonic development is a critical process during which the embryo establishes a close relationship with maternal uterus, forms the basic body plan, and differentiates into three germ layers (endoderm, mesoderm and ectoderm) through gastrulation1. The endoderm gives rise to most of the internal linings of the embryo body, including the lungs, the liver, the pancreas and the intestinal tract; the mesoderm differentiates into the heart, the skin, the muscle system and the bones; the ectoderm forms the nervous systems, the mammary glands and the hair. The abnormal development of human peri-implantation embryos appears to be correlated with the frequencies of woman reproductive disorders and human major structural malformations in central nervous system, heart and limbs2. However, the knowledge of human peri-implantation embryonic development and gastrulation is very limited.
Peri-implantation embryonic development and gastrulation have been very difficult to observe and analyze, since these events happen within the uterus. Thus, in vitro culture (IVC) systems may serve as a powerful tool to solve this problem. Though significant advances have been made in murine development with mouse embryo IVC systems, early embryogenesis in the rodent is distinct from that in primates3, 4. In 2016, Zernicka-Goetz’s lab and Brivanlou’s lab cultured human embryos in vitro for 12-13 days, leading to extensive discussions worldwide5, 6. Because many governments and international organizations have recommended that human embryos should not be allowed to grow beyond 14 days in vitro, a non-human primate embryo IVC system is urgently needed to understand the gastrulation of early primate embryogenesis, and the related problems and diseases that arise during early human development.
On October 31st, 2019, a research article entitled “In vitro culture of cynomolgus monkey embryos beyond early gastrulation” was published in Science. This work was done by three teams: Dr. Hongmei Wang’s group and Dr. Lei Li’s group from the Institute of Zoology of the Chinese Academy of Sciences (CAS) / the Innovation Academy for Stem Cell and Regeneration of CAS, and Dr. Ping Zheng’s group from the Kunming Institute of Zoology of CAS. In this study, the authors report the establishment of an IVC system which supported the continuous development of monkey embryos beyond early gastrulation and to 20 days post fertilization (d.p.f). Firstly, these authors optimized an IVC system for the culture of mouse blastocysts beyond early postimplantion stage. Then, they used this optimized system to culture cynomolgus monkey embryos to 20 days. At d.p.f. 13-14, a bilaminar disc-like structure appeared in ~27.7% of the IVC embryos. At d.p.f. 15-16, the disc-like structure was clearly observable in the IVC embryos under the optical microscope. Some of the IVC embryos successfully developed to d.p.f. 20. Histological and immunofluorescent staining results demonstrated that the IVC embryos recapitulated the key events of in vivo early embryo development, including segregation of the epiblast and hypoblast, formation of the amniotic and yolk sac cavities, appearance of the primordial germ cells and the initiation of gastrulation. Furthermore, single-cell RNA-seq analyses showed that the IVC embryos were similar to their in vivo counterparts in gene expression profiles and cell types. In addition, this study revealed the molecular signatures of several cell types of primate early post-implantation embryos, including amnion cells.
Altogether, this study provides compelling evidence supporting that the cynomolgus monkey embryos can develop beyond early gastrulation and to 20 days post fertilization in their IVC system. Furthermore, this study provides novel information on cell lineage specification during primate early post-implantation development. In combination with CRISPR-Cas9-mediated gene editing and cell lineage tracing, this system will accelerate our understanding of the mysterious dynamics of early embryonic development in primates, with possible relevance to human early embryonic development and diseases.
The study was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences, the National Key R&D Program of China, the National Natural Science Foundation of China and the exchange program of State Key Laboratory of Genetic Resources and Evolution, Kunming Institute of Zoology, Chinese Academy of Sciences.
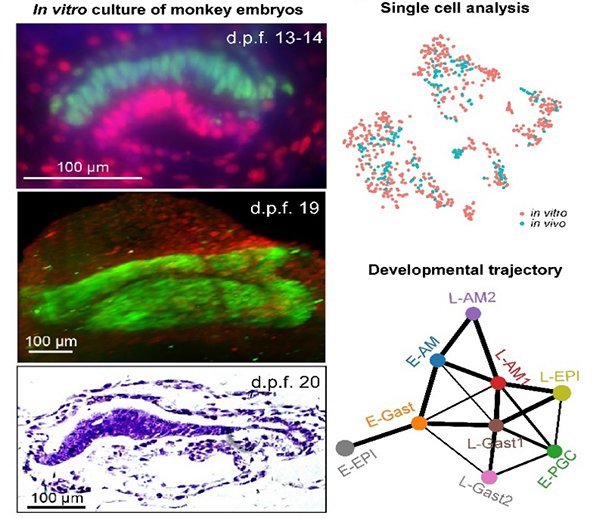
Monkey embryos grow in vitro beyond early gastrulation. The in vitro cultured embryos were stained with the antibodies for OCT4 (green) and GATA6 (red), and hematoxylin and eosin staining. Single-cell transcriptome analysis revealed the similarities among cell types and developmental trajectory of epiblast derivatives in the in vitro and in vivo monkey early embryos.
Article link:
1. Arnold, S.J. & Robertson, E.J. Making a commitment: cell lineage allocation and axis patterning in the early mouse embryo. Nat Rev Mol Cell Biol 10, 91-103 (2009).
2. Rossant, J. & Tam, P.P.L. Exploring early human embryo development. Science 360, 1075-1076 (2018).
3. Bedzhov, I. & Zernicka-Goetz, M. Self-Organizing Properties of Mouse Pluripotent Cells Initiate Morphogenesis upon Implantation. Cell 156, 1032-1044 (2014).
4. Nakamura, T. et al. A developmental coordinate of pluripotency among mice, monkeys and humans. Nature 537, 57-62 (2016).
5. Deglincerti, A. et al. Self-organization of the in vitro attached human embryo. Nature 533, 251-4 (2016).
6. Shahbazi, M.N. et al. Self-organization of the human embryo in the absence of maternal tissues (vol 18, pg 700, 2016). Nature Cell Biology 18 (2016).

Dr. Nicolas PlachtaHHMI investigatorSenior Principal Investigator, IMCB, A*STAR, SingaporeThis method opens a completely new avenue to investigate primate gastrulation in real time. Combined with the power of modern microscopy techniques, the system reported by Ma, Wang and colleagues will enable the discovery of key processes underlying primate gastrulation, which can not be easily captured using fixed specimens or analyzing other non-primate species. The work can have profound effects for understanding developmental abnormalities resulting from gastrulation defects, and also generally promote research using monkey models. Although logistically more challenging than other animal models, the advantages of these new types of methods will likely make research on non-human primates considerably more appealing.

Dr. Magdalena Zernicka-Goetz, Ph.D., ProfessorUniversity of Cambridge, UKFellow of the United Kingdom Academy of Medical Sciences (FMedSci)It is very exciting. Monkey embryos were previously cultured only up to blastocyst stage (day 7). I am delighted that our method for culturing human embryos was useful here and can be adopted further. Perhaps the most surprising finding for me is that the authors were able to culture the embryos up to day 20 (beyond gastrulation). True achievement as now there is a system to study gastrulation in vitro in a model very similar to the human embryo.



