In human, the skin is one of the organs that exhibit early-onset aging-associated dysfunction. As a critical physical barrier, the skin is composed of a variety of cell types including fibroblasts and keratinocytes. Due to its high heterogeneity, it is difficult to accurately reveal the cell type-specific molecular mechanisms of human skin aging, which hinders our understanding of human skin aging and the development of interventions for human skin aging and related diseases.
Recently, scientists from the Institute of Zoology of the Chinese Academy of Sciences and Beijing Institute of Genomics of the Chinese Academy of Sciences have worked jointly and established the first single-cell transcriptomic atlas of human skin aging. Using the single-cell transcriptome sequencing technology, they drew a comprehensive aging roadmap of human skin at the single-cell resolution and identified the disturbance of growth-controlling transcription factors as a major feature of human skin aging. The study entitled "A Single-Cell Transcriptomic Atlas of Human Skin Aging" was published online in Developmental Cell on November 25rd, 2020.
The researchers first obtained eyelid skin samples from a group of healthy female donors of different ages. Morphological analysis revealed that epidermal thickness, the convolution of dermal-epidermal junction and dermal collagen density were decreased during aging. Through the single-cell transcriptome sequencing analysis, the researchers identified 11 skin cell types with unique gene expression signatures and classified human epidermal basal cells into six subpopulations of quiescent or amplifying epidermal basal cells based on their transcriptional identities by hierarchical differentiation.
An in-depth dissection of the changes of gene expression during human skin aging revealed that the skin of middle-aged individuals was transcriptionally more similar to the skin of elderly individuals than to the skin of young individuals, indicating that age-related gene expression changes start to accumulate early in life. Compared with younger individuals, the skin of elderly individuals exhibited increased inflammation and decreased epithelial maintenance ability. As the eyelid skin was exposed to daily sunlight, further analysis revealed age-related decrease in DNA repair capacity and increase in biomacromolecular damage in several types of skin cells, suggesting that chronic inflammation and photodamage may be important triggers of ocular skin aging.
In addition, the researchers observed early-onset downregulation of growth-controlling transcription factors (HES1 in fibroblasts and KLF6 in basal cells) underlying the cell type-specific transcriptional changes. Consistently, inactivation of HES1 in human dermal fibroblasts or KLF6 in human keratinocytes led to cellular senescence. Furthermore, activation of HES1 alleviated the senescence of dermal fibroblasts, indicating that HES1 may exert protective effects on the skin. Finally, the researchers found that a natural compound quercetin attenuated the senescence of dermal fibroblasts, providing a new potential intervention strategy for interfering human skin aging.
Taken together, this study provides an in-depth understanding of the cell type-specific mechanisms of human skin aging at the single-cell level and thus facilitates the development of novel therapeutic strategies combating aging-related skin diseases. In addition, this study has established a discovery pipeline for small-molecule compounds against human skin aging. The data of this study have been uploaded to the Aging Atlas (https://bigd.big.ac.cn/aging/landscape?project=Human_Skin).
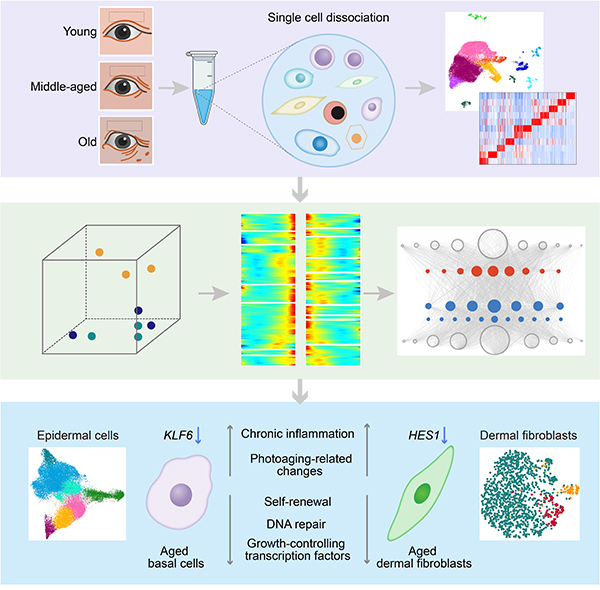
Figure. Single-Cell Transcriptomic Analysis of Human Skin Aging
(Contact: Liu Guanghui, ghliu@ioz.ac.cn)

