Regeneration is an important process of rejuvenating or replacing damaged, diseased, or aged tissues. Regenerative capacity declines with evolution and age. For example, salamanders in lower animals have the complete regenerative capacity in limbs, while in most mammals including humans, limited regeneration and functional recovery capabilities reside in young tissues and decline with age. To date, the molecular mechanism underlying the declined regenerative capacity with evolution and age remains poorly understood. Unlike proteins that are biomacromolecules, the structure of metabolites is relatively similar between species, which makes metabolism an ideal research area for studying evolutionarily conserved biology. Yet, little is known about the small-molecule metabolites that potentially regulate aging and regeneration processes.
Recently, researchers from the Institute of Zoology and Beijing Institute of Genomics of the Chinese Academy of Sciences have collaborated to identify uridine as a pro-regenerative metabolite that promoted human stem cell activity and enhanced regeneration and tissue repair in multiple tissues in mammals. This study entitled “Cross-species metabolomic analysis identifies uridine as a potent regeneration promoting factor” was published online in Cell Discovery on February 1st, 2022.
In this study, by combining metabolomics and transcriptomics approaches, the researchers revealed several cross-species and cross-ages metabolic programs associated with higher regenerative capacity, i.e., polyamine metabolism, pyrimidine metabolism (uracil containing), and fatty acid metabolism pathways. Combined with the human stem cell aging research system, the researchers conducted screening for natural metabolite candidates with regeneration-promoting potentials and identified that uridine counteracted cellular senescence in both physiologically and pathologically senescent cells. Next, the researchers found that uridine supplementation promoted regeneration or repair of various tissues in vivo, including skeletal muscle, heart, liver, skin, and articular cartilage. Specifically, in the muscle injury model, uridine effectively improved muscle regeneration and repair, alleviated the inflammatory response, and endowed uridine-treated mice with higher grip strength and locomotive activity. In the myocardial infarction model, uridine ameliorated acute inflammation and improved the contractility of the injured heart. In the liver fibrosis model, uridine alleviated carbon tetrachloride-induced liver fibrosis and restored the liver function. In the hair regeneration model, uridine induced anagen hair growth. In the arthritis model, uridine promoted the regeneration of articular cartilage and improved grip strength and locomotive activity of mice. In addition, researchers also found that uridine was more abundant in the plasma from young individuals than that from old individuals. Additionally, oral uridine supplementation improved the physical activities of physiologically aged mice.
Taken together, the researchers proved that the supplementation of single metabolite uridine alleviated stem cell senescence, promoted tissue regeneration and repair, and improved the physical activities of aged mice. This study provides previously unknown metabolism-associated regeneration principles across species and opens new avenues for metabolic intervention in tissue repair, regeneration, and aging.
Link: https://www.nature.com/articles/s41421-021-00361-3
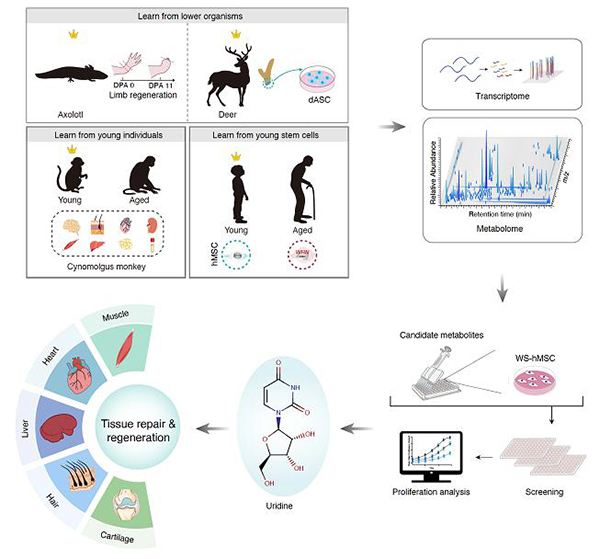
Figure legend: Identification of key metabolic small molecules that promote multi-tissue repair and delay aging through cross-species and cross-age metabolome profiling
(Contact: Guang-Hui Liu, ghliu@ioz.ac.cn)

