The spinal cord, a vital bridge connecting the brain and peripheral nerves, plays a pivotal role in governing motor functions and coordinating basic life activities of the body, which mainly through a rare and critical group of cells within the spinal cord called motor neurons.
As the "command center" for human movement and basic organ autonomous activities, the spinal cord's aging process holds significant implications for the coexistence of various chronic diseases in the elderly population. However, the mechanisms of spinal cord aging remain poorly understood. What biomarkers can we use to measure spinal cord aging? What drives spinal cord aging? Can interventions be developed to alleviate spinal cord aging?
Addressing the knowledge gap surrounding spinal cord aging, researchers from the Institute of Zoology and Beijing Institute of Genomics, both affiliated with the Chinese Academy of Sciences, have achieved an original discovery. Their study, entitled “CHIT1-positive microglia drive motor neuron aging in the primate spinal cord”, published online in Nature on October 31st, 2023, unveiled a previously unknown subtype of CHIT1-positive microglia in the aged spinal cord of non-human primates (Figure 1). These cells, which they coined the name AIMoN-CPM (Aging-Induced Motor Neuron toxic CHIT1-Positive Microglia), play a driving role in motor neuron aging by activating SMAD signaling through the secretion of the CHIT1 protein.
Metaphorically speaking, microglia function similarly to security personnel in the spinal cord. However, with age, some of them become excessively vigilant, accelerate the aging process of motor neurons. Promisingly, the study highlights that blocking the CHIT1-SMAD signaling pathway or supplementing with vitamin C can counteract the pro-aging effects of this “overzealous security” on spinal cord motor neurons.
Moreover, elevated CHIT1 levels were observed in both the cerebrospinal fluid and blood of elderly individuals compared to younger counterparts. This fluid-based biomarker holds potential for assessing the degree of spinal cord aging.
“This study provides the first systematic characterization of phenotypic, pathological, cellular and molecular features of primate spinal cord aging, deepening our understanding of human spinal cord degeneration,” said Prof. Guang-hui Liu, the lead investigator of this study. “By targeting AIMoN-CPM and CHIT1 signaling, we hold promise for slowing down spinal cord aging and managing age-related comorbidities.
This research sheds light on the intricate mechanisms underlying spinal cord aging, offering hope for the development of interventions to alleviate its effects. As the aging population continues to grow, this study paves the way for potential strategies to enhance quality of life and manage age-related health challenges.
Link: https://www.nature.com/articles/s41586-023-06783-1
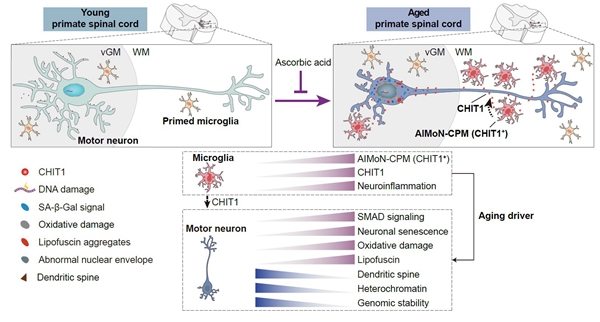
Figure. Panoramic view of primate spinal cord aging (image by Shuhui Sun)
Contact:
Guang-Hui Liu
Institute of Zoology Chinese Academy of Sciences
Tel: 86-64807852
E-mail: ghliu@ioz.ac.cn
Web: http://english.ioz.cas.cn/

